News
20251202
Congratulations to Chunsong Lu on obtaining his doctoral degree!
We are delighted to announce that Chunsong Lu has successfully obtained his doctoral degree! His innovative work on MOF-derived materials offers promising new approaches for addressing water pollution challenges. His dedication and contributions during his time in the group have been truly appreciated.
We wish him continued success and fulfillment in all his future endeavors!

20250922
Great news! Qiuning Li has won the Excellent Paper Award at the 2025 China Mass Spectrometry Academic Conference!

20250912
Congratulations to Mingying Shi for winning the Best Poster Award at BCEIA 2025!

20250625
Daiyu Miao Presents an Oral Presentation at the Young Scientist Forum of AOMSC 2025 in Okinawa, Japan.
Daiyu Miao from our research group attended the 10th Asia-Oceania Mass Spectrometry Conference (AOMSC) in Okinawa, Japan, where he presented his latest research on live cell labeling through an oral presentation. During the conference, he shared key findings and advancements in the field with fellow researchers.

20250529
We warmly congratulate Yi Zhang on receiving her Ph.D. degree, and welcome her to continue her postdoctoral research in our group.

2025.5.17
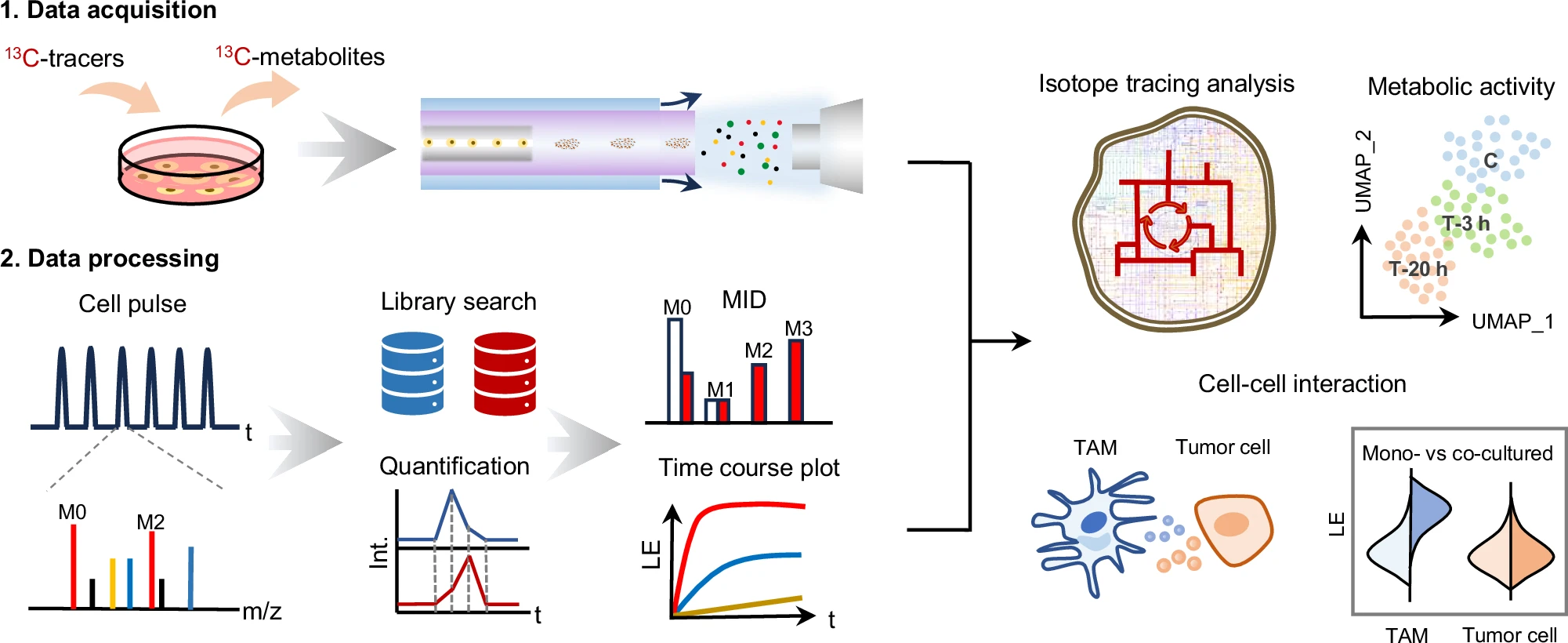
Congratulations to Yi Zhang on the successful publication of her paper "Dynamic single-cell metabolomics reveals cell-cell interaction between tumor cells and macrophages" in Nature Communications.
Abstract
Single-cell metabolomics reveals cell heterogeneity and elucidates intracellular molecular mechanisms. However, general concentration measurement of metabolites can only provide a static delineation of metabolomics, lacking the metabolic activity information of biological pathways. Herein, we develop a universal system for dynamic metabolomics by stable isotope tracing at the single-cell level. This system comprises a high-throughput single-cell data acquisition platform and an untargeted isotope tracing data processing platform, providing an integrated workflow for dynamic metabolomics of single cells. This system enables the global activity profiling and flow analysis of interlaced metabolic networks at the single-cell level and reveals heterogeneous metabolic activities among single cells. The significance of activity profiling is underscored by a 2-deoxyglucose inhibition model, demonstrating delicate metabolic alteration within single cells which cannot reflected by concentration analysis. Significantly, the system combined with a neural network model enables the metabolomic profiling of direct co-cultured tumor cells and macrophages. This reveals intricate cell-cell interaction mechanisms within the tumor microenvironment and firstly identifies versatile polarization subtypes of tumor-associated macrophages based on their metabolic signatures, which is in line with the renewed diversity atlas of macrophages from single-cell RNA-sequencing. The developed system facilitates a comprehensive understanding single-cell metabolomics from both static and dynamic perspectives.


2025.1.8
Congratulations to Shaojie Qin on the publication of his paper "Multi-dimensional bio mass cytometry: simultaneous analysis of cytoplasmic proteins and metabolites on single cells" in Chemical Science.
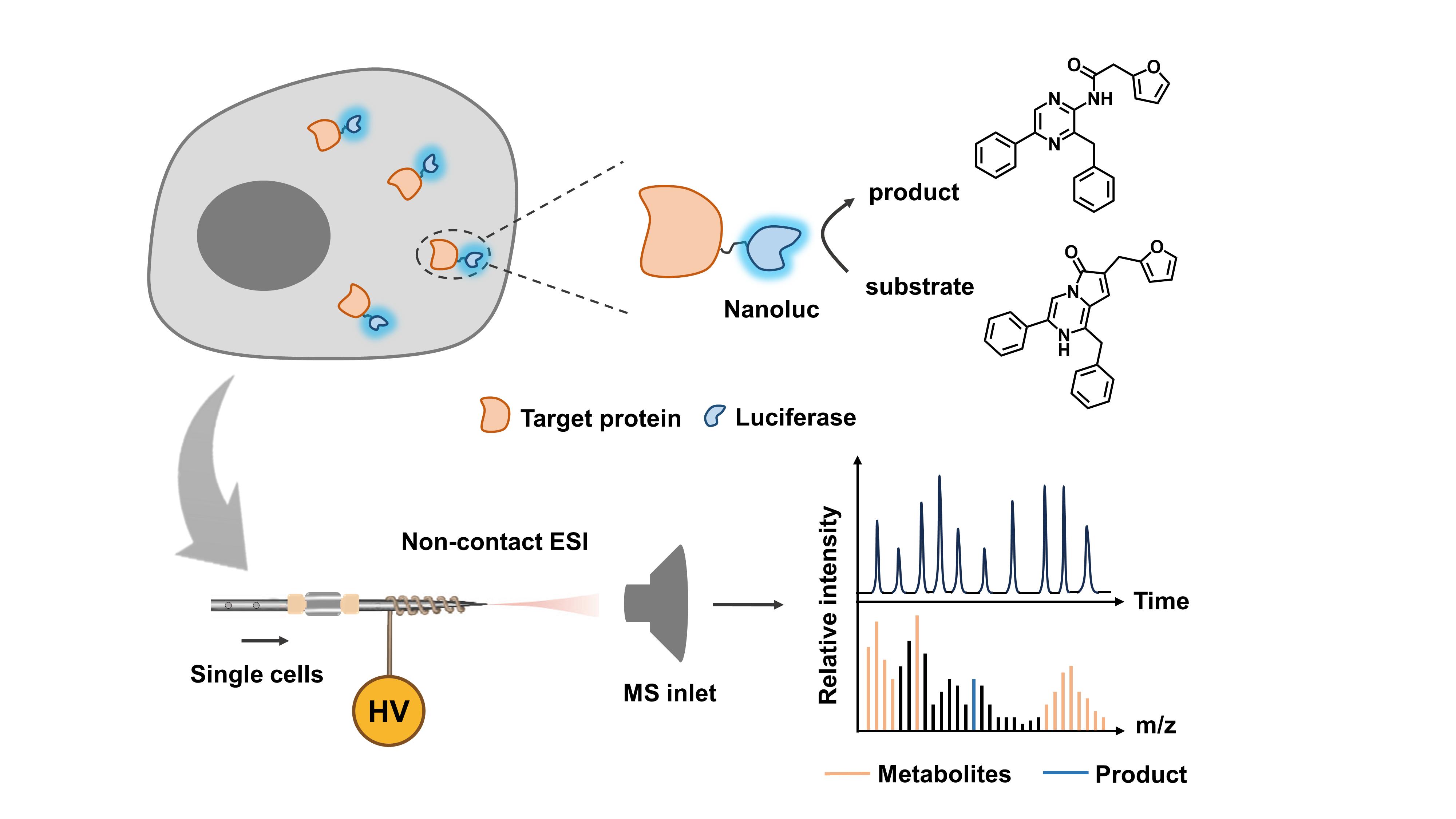
Abstract
Single-cell multi-dimensional analysis enables more profound biological insight, providing a comprehensive understanding of cell physiological processes. Due to limited cellular contents, the lack of protein and metabolite amplification ability, and the complex cytoplasmic environment, the simultaneous analysis of intracellular proteins and metabolites remains challenging. Herein, we proposed a multi-dimensional bio mass cytometry platform characterized by protein signal conversion and amplification through an orthogonal exogenous enzymatic reaction. Clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated protein 9 (Cas9) gene editing technology was applied in the quantification of endogenous intracellular protein glycer-aldehyde-3-phosphate dehydrogenase (GAPDH) through exogenous luciferase Nanoluc (Nluc). The simultaneous detection of GAPDH and hundreds of metabolites at the single-cell level was realized for the first time. Semiquantitative analysis of GAPDH together with single-cell metabolomes under S-nitrosoglutathione (GSNO)-induced oxidative stress was investigated. Bioinformatics analysis revealed 16 metabolites that correlated positively with GAPDH expression upon oxidative stress, including long-chain fatty acids (palmitoleic acid, myristic acid, etc.) and UDP-N-acetylglucosamine (UDP-GlcNAc). Potential synergetic functions of GAPDH and UDP-GlcNAc-mediated oxidative stress responses were also elucidated. Our work proposes a novel strategy for the simultaneous quantitative analysis of single-cell intracellular proteins and metabolites, deepens the understanding of inherent anti-oxidative stress response mechanisms, and provides the molecular fundamentals for the study of inherent biological processes.
2024.10.23
Congratulations to Shaojie Qin and Caiwei Hu for winning the Best Youth Oral Presentation Award and Best Poster Award, respectively, at HPLC 2024!

2024.6.5
Congratulations to Shaojie Qin on obtaining his doctoral degree! We also extend our warmest congratulations to Dr. Xue Zhang and Dr. Xiaodan Qiu on their successful completion of their postdoctoral programs!



2024.6.20
Yi Zhang Attends Metabolomics 2024 in Osaka, Japan.
Yi Zhang from our research group attended Metabolomics 2024 in Osaka, Japan, where she presented her latest metabolomics research via a poster and discussed her key findings and breakthroughs in the field.
2024.5.23
Congratulations to Shaojie Qin on the successful publication of his paper in Nature Communications!
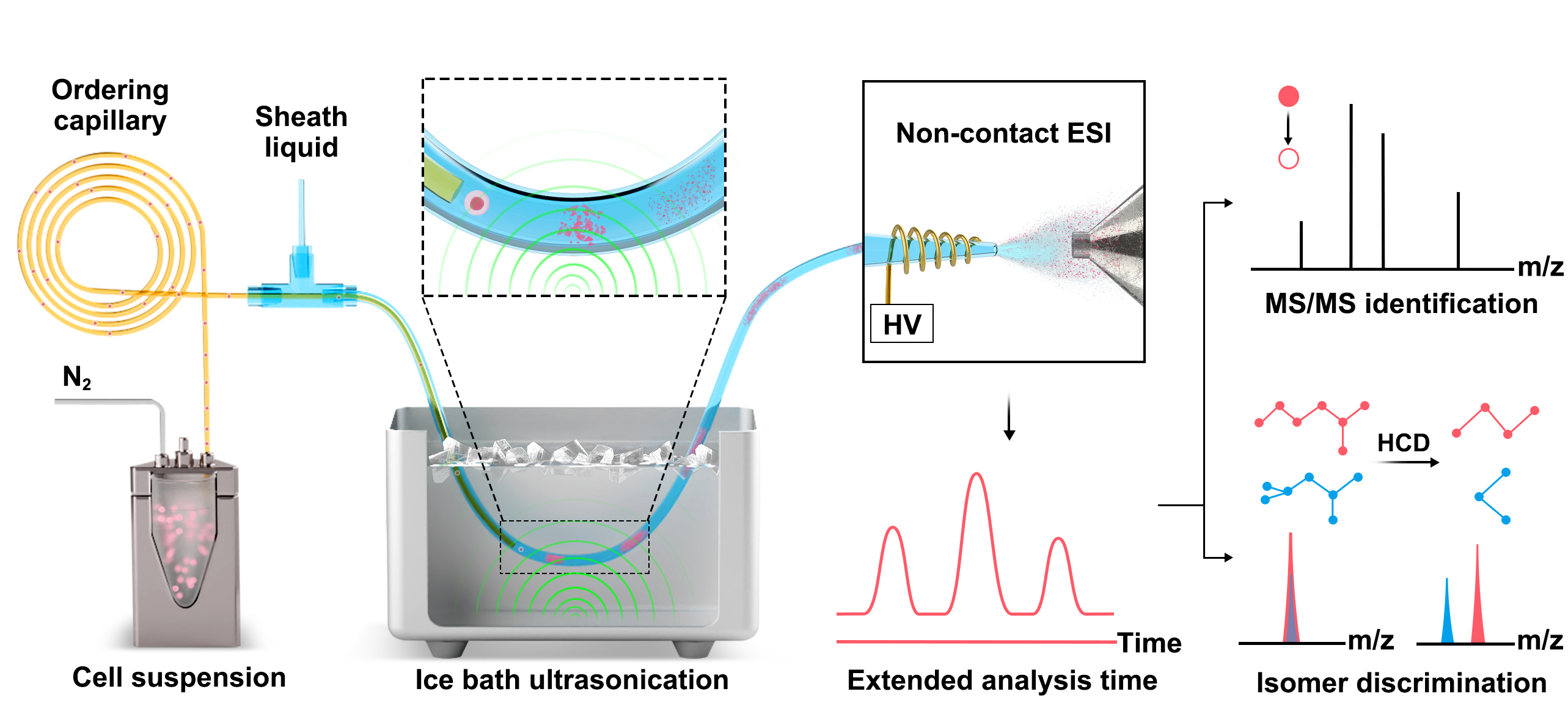
2024.3.20
Congratulations to Yi Zhang on the publication of her review article!
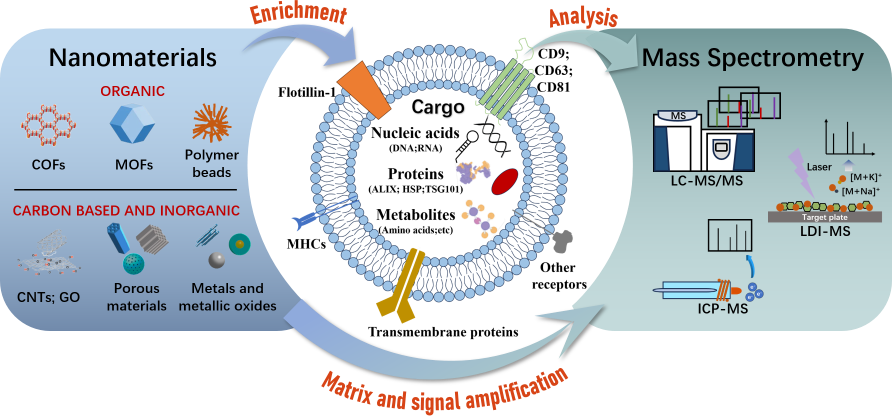
Abstract
Single-cell multi-dimensional analysis enables more profound biological insight, providing a comprehensive understanding of cell physiological processes. Due to limited cellular contents, the lack of protein and metabolite amplification ability, and the complex cytoplasmic environment, the simultaneous analysis of intracellular proteins and metabolites remains challenging. Herein, we proposed a multi-dimensional bio mass cytometry platform characterized by protein signal conversion and amplification through an orthogonal exogenous enzymatic reaction. Clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated protein 9 (Cas9) gene editing technology was applied in the quantification of endogenous intracellular protein glycer-aldehyde-3-phosphate dehydrogenase (GAPDH) through exogenous luciferase Nanoluc (Nluc). The simultaneous detection of GAPDH and hundreds of metabolites at the single-cell level was realized for the first time. Semiquantitative analysis of GAPDH together with single-cell metabolomes under S-nitrosoglutathione (GSNO)-induced oxidative stress was investigated. Bioinformatics analysis revealed 16 metabolites that correlated positively with GAPDH expression upon oxidative stress, including long-chain fatty acids (palmitoleic acid, myristic acid, etc.) and UDP-N-acetylglucosamine (UDP-GlcNAc). Potential synergetic functions of GAPDH and UDP-GlcNAc-mediated oxidative stress responses were also elucidated. Our work proposes a novel strategy for the simultaneous quantitative analysis of single-cell intracellular proteins and metabolites, deepens the understanding of inherent anti-oxidative stress response mechanisms, and provides the molecular fundamentals for the study of inherent biological processes.
2023.8.17
Congratulations to Shaojie Qin for winning the Oral Presentation Prize at the ANLY Virtual Asia-Pacific Graduate Student Symposium, ACS Fall 2023!