Ischemic stroke is a leading cause of disability and death worldwide. Oxidative stress is one of the hallmarks of ischemic stroke, leading to the dysfunction/death of neurons. Catalase-based (CAT) biomimetic complexes are emerging as promising therapeutic candidates that are expected to act as neuroprotectants for ischemic stroke by decreasing the damaging effects from H2O2. Note that this process is in direct competition with unwanted production of the harmful hydroxyl radical, HO•. Thus, an ideal therapeutic CAT antioxidant biomimetic should not only promote effective H2O2 dismutation but also minimize the formation of HO•.
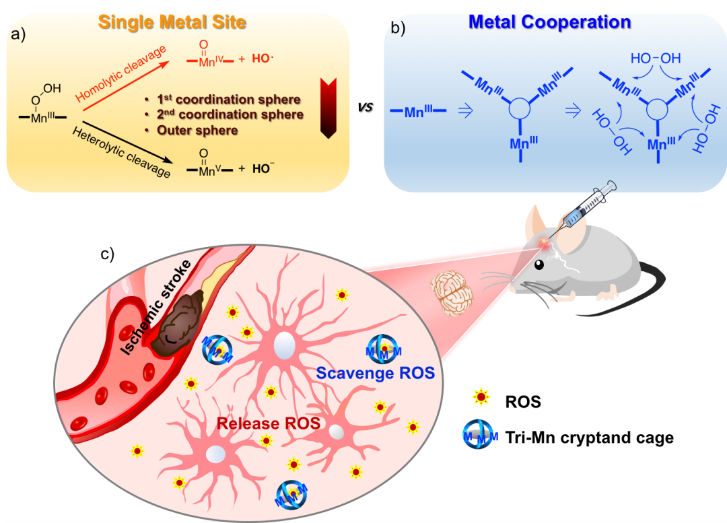
Figure 1. Schematic illustration of (a) homolytic and heterolytic O–O bond cleavage of an edge-bound Mn(III) peroxide complex, (b) the effect of metal cooperativity in binding and splitting H2O2, and (c) the strategy developed in the present study.
Toward this goal, Jun-Long Zhang’s group, college of chemistry and molecular engineering (CCME), Peking University (PKU), collaborated with Song Gao’s group (CCME), Lei Kang’s group (First Hospital, PKU), Jonathan Sessler’s group (U. Texas, Austin), has reported a metal cooperative antioxidant strategy that overcomes ischemic stroke damage. This original experimental and intellectual work was published in Journal of American Chemical Society (2020, 142, 10219-227), entitled “Tri-Manganese(III) Salen-Based Cryptands: A Metal Cooperative Antioxidant Strategy that Overcomes Ischemic Stroke Damage In Vivo”
Within the context of ongoing efforts to explore potential biological applications of biomimetic metalloenzyme complexes, they reasoned that metallotris (Salen) cryptands could also provide a platform for the cooperative binding of guest molecules as the result of the metal centers being in close proximity. The study demonstrated an overall 6.8-fold increase in oxygen production for trinuclear complexes relative to mononuclear complex on a per mole basis. More importantly, comparison of the production of hydroxyl radical by integration of the EPR signals and fluorescence titration clearly showed the trinuclear complex produced minimal HO•. Further CAT activity assay and mechanistic studies supported a metal cooperative effect by stabilizing H2O2 between neighboring Mn(IV)═O moieties, resulting in an improved CAT activity, thus providing an expected therapeutic benefit.

Figure 2. (a) Oxygen production over time (s) via catalytic H2O2 (10 mM) dismutation. (b) Maximum concentration of oxygen (at 1000 s) produced via the catalytic H2O2 dismutation process in (a). (c) Fluorescence emission spectra of p-phthalic acid (5 mM). (d) X-band EPR spectra. (e) X-band EPR spectra. (f) Changes in the UV–vis spectra seen when Mnsalen was treated with 3 equiv of H2O2 in 50 mM PBS buffer (pH = 6) at 5 °C.
On the basis of an in vivo stroke model, they concluded that the neurological function of rats was improved in a statistically significant manner (P < 0.01;) when treated with trinuclear complex relative to the control group following ischemia. Relative to the mono-Mn complex, the tri-Mn(Salen) system provided a statistically significant greater therapeutic benefit (P value <0.01), as judged by a number of indicia. Fluorescence imaging assays and immunohistochemical staining provided evidence to support the notion that the neuroprotective effect resulted from reduction in the disease model-associated ROS concentrations, which in turn lead to reduced apoptosis of nerve cells and lower expression levels of apoptosis-associated enzymes.

Figure 3. (a) Schematic of the stroke operation and injection procedure. (b) Biodistribution of 1 detected by MRI (7 T) after the surgery in (a). (c) PET images of rat brain following administration (tail vein injection) of 18F-FDG to animals. (d) Coronal brain sections of rats (stained with triphenyltetrazolium chloride (TTC) solution). (e) Calculated infarct volumes under the different conditions in (d). **P < 0. 01, ***P < 0.001.
This work highlights how multicentered manganese complexes, if appropriately designed, might have a role to play in the treatment of diseases related to oxidative stress. On the other hand, this study also provides a new research paradigm for biomimetic inorganic chemistry, that is accessible for the design of metal drugs, to explore the potential applications in disease diagnostic and therapy. Yingying Ning is the first author of the paper. Jun-Long Zhang, Lei Kang and Jonathan Sessler are co-corresponding authors. Other collaborators include Gao Song, Wang Bingwu and Jiang Shangda. Recently, the ChemistryViews.org website promoted the work under the title "Manganese Complex Protects against H2O2-Induced Stroke Damage". The research was funded by the National Natural Science Foundation of China, the Ministry of Science and Technology, and the Beijing National Research Center for Molecular Sciences.